As a follow-up to my controversial post on the effect of infrared “back radiation” downwelling from the colder sky to the warmer surface, the existence of which some dispute (despite the real-time availability of such data), I’ve come up with an experimental setup to see how IR radiation from the sky influences air temperature near the ground. (Yes, I know some of you think there is no such thing, but please indulge my fantasy as if it was true, ok?)
The design is pretty simple and inexpensive, and looks a little like the blackbody radiators that are used as calibration sources. The following cartoon shows the main components:
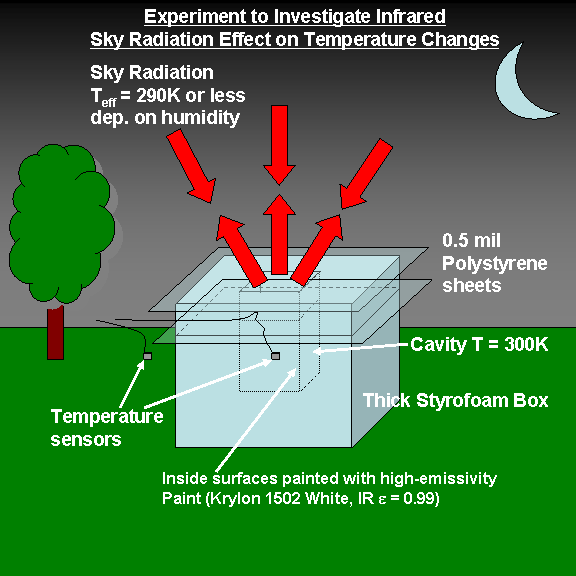
The idea is to isolate a sample of air and control its environment so that it’s main source of energy gain or loss is through an opening that looks at the sky. You have probably noticed that on a clear evening, dew forms first on the tops of cars and other surfaces. This is because these surface are losing IR energy faster than the air and other surfaces are, so their temperature falls below the dewpoint temperature first.
If we can isolate that effect sufficiently from other sources and sinks of energy, we should be able to get air temperature drops within the cavity in the direction of the colder, effective sky temperature. (We use air since it is very hard to measure the temperature of a cold surface accurately, so we let the cold surface inside the cavity chill the air in contact with it).
The cavity will be lined with aluminum foil, which has very high reflectivity in the infrared, painted on the inside only with high-emissivity paint (Krylon flat white, #1502 if I can find it…apparently, black paint isn’t as good an emitter in the IR.)
The 2 thin polyethylene sheets are in the upward-looking cavity opening to trap a layer of air for thermal conductive insulation, while at the same time passing most IR radiation (something polyethylene is apparently quite good at). The thermal conductivity of the trapped air is a little better (less) than that of Styrofoam, but since convection can occur in an air cavity, I’m sure the actual rate of heat transfer will be more than that for Styrofoam.
SO WHAT KIND OF SIGNALS CAN WE EXPECT?
(…assuming the experiment isn’t a complete failure because of something important I haven’t thought of…)
If you search around on the internet you will find that those who have made such broadband IR measurements of the sky (from what I can tell, usually with instruments that measure between 8 and 14 microns wavelength) report that the effective sky temperature in the infrared is usually 10 to 30 deg. C lower than the near-surface air temperature. Ten deg. C is more typical during humid conditions or cirrus cloud cover, while 30 deg. C would be during clear, low humidity conditions.
Low clouds produce a downwelling sky temperature nearly the same as the upwelling temperature. The sky temperature increases as you scan from the zenith down in elevation, due to the greater path length through the atmosphere.
As an example of the theoretically-expected difference in IR energy flows in and out of the cavity, at an emissivity of 1, a cavity at 300 K temperature should emit a broadband IR flux of 459 Watts/m2, while a downwelling apparent temperature of 290 K (10 deg. lower than the cavity) would produce 401 Watts/m2, the difference being 58 Watts per sq. meter.
In a perfect setup with a cavity emissivity of 1 and no other losses of energy under these conditions, the inside of the cavity would then cool to 10 deg. C less than the surrounding air temperature as the insulated cavity comes into radiative equilibrium with the sky. (I am currently monitoring 2 temperatures in my back yard, with the data sent to my computer by wireless. My first design failed due to large conductive energy loses, which led to the 2nd design, above).
Of course, a “perfect” experimental setup is not possible. I’ve run some numbers based upon the thermal conductivity of Styrofoam and I think I can keep the energy loses to about 20% of the signal being sought, but this is uncharted territory for me.
OK, TIME FOR YOUR PREDICTIONS
So, for all of you who think you know what will happen in this experiment, come on and tell the rest of us. Will the temperature of the air in the cavity stay the same? Will it cool? By how much?
I especially want to hear an answer to 2 questions:
(1) If you think the cavity will be the only source of IR radiation, and there is no downwelling IR radiation from the sky, then what will keep the air temperature inside from falling dramatically lower than the air temperature outside of the box?
(2) If you think the temperature in the cavity will not change, then what is keeping the IR radiation flowing out of the cavity toward the sky from causing a temperature fall? Wouldn’t want to violate the 1st Law of Thermodynamics, ya know.
Let the thinking begin.
 |

 Home/Blog
Home/Blog



